Image Wisely, a joint initiative of ACR, RSNA, ASRT and AAPM,
provides information to the medical community to promote safety in medical imaging.

Optimizing Oncologic FDG-PET/CT Scans to Decrease Radiation Exposure
Esma A. Akin, MD, George Washington University Medical Center, Washington, DC
Drew A. Torigian, MD, MA, University of Pennsylvania Medical Center, Philadelphia, PA
Patrick M. Colletti, MD, University of Southern California Medical Center, Los Angeles, CA
Don C. Yoo, MD, The Warren Alpert Medical School of Brown University, Providence, RI
Download PDF

The development of dual-modality positron emission tomography/computed tomography (PET/CT) systems with near-simultaneous acquisition capability has addressed the limited spatial resolution of PET and has improved accurate anatomical localization of sites of radiotracer uptake detected on PET. PET/CT also allows for CT-based attenuation correction of the emission scan without the need for an external positron-emitting source for a transmission scan. This not only addresses the limitations of the use of noisy transmission data, therefore improving the quality of the attenuation-corrected emission scan, but also significantly decreases scanning time. However, this comes at the expense of increased radiation dose to the patient compared to either PET or CT alone.
Optimizing PET protocols
The radiation dose results both from the injected radiotracer 18F-2-fluoro-2-deoxy-D-glucose (FDG), which is ~7 mSv from an injected dose of 10 mCi (given the effective dose of 0.019 mSv/MBq (0.070 rem/mCi) for FDG), as well as from the external dose of the CT component, which can run as high as 25 mSv [1]. This brings the total dose of FDG-PET/CT to a range of ~8 mSv up to 30 mSv, depending on the type of study performed as well as the anatomical region and number of body parts imaged, although several recent studies have reported a typical average dose of ~14 mSv for skull base-to-thigh FDG-PET/CT examinations [2-8]. The critical organ after FDG administration is the urinary bladder, which is exposed to 0.16 mGy/MBq (0.59 rad/mCi) in adults, although this can be reduced with patient hydration and increased patient voiding frequency [1].
Reduction of FDG dose with the use of currently available PET/CT systems can be challenging due to the short half-life of FDG (109.8 min) and limitations imposed by patient size. Larger patients may have resultant images with lower signal-to-noise ratios and reduced image quality. For significantly heavy patients (>90 kg), increase of scanning time (time per bed position) rather than increase of FDG activity is preferable to improve image quality without increasing dose [9]. Interestingly, a recent report suggests that FDG dose can be reduced by 50% without a loss of diagnostic performance in the setting of whole-body PET/CT for the cancer patient [10]. (See Table 1 for a summary of methods to decrease dose.) Further research will be required to determine how low the requisite FDG dose can be reduced for use with currently available PET/CT instrumentation without compromising diagnostic quality.
Newer imaging units with faster crystals produce higher light output, which may be used to shorten exam times or to allow use of lower doses of FDG. Time-of-flight (TOF) information in image reconstruction also appears promising in the ongoing efforts to reduce dose [7, 11, 12]. TOF can pinpoint the origination of positron annihilation more accurately compared to non-TOF reconstruction. This improves the signal-to-noise ratio, reduces image degradation due to attenuation and scatter, and can improve image quality in heavy patients without having to increase injected dose [13, 14]. The 3D PET emission acquisition mode also allows for a reduction in injected dose by up to 50% relative to the recommended FDG dose for the 2D mode of acquisition [1, 15].
| TABLE 1 - METHODS TO DECREASE RADIATION EXPOSURE IN FDG-PET-CT | |
| Optimize utilization of FDG-PET-CT | Optimize protocols to reduce dose while maintaining sufficient image quality |
|
Perform FDG-PET-CT only when clinically indicated
|
PET-related methods to reduce dose
|
| Use alternative non-ionizing radiation imaging technologies (US, MRI) whenever possible |
CT-related methods to reduce dose
|
| Consider use of PET-MRI in place of PET-CT for certain clinical applications to reduce dose, although more research data is needed | |
| Perform routine quality assurance and quality control of imaging instrumentation and optimization of imaging protocols | |
| Monitor patient dose exposure from individual imaging examinations and on a cumulative basis | |
| Monitor patient dose exposure from individual imaging examinations and on a cumulative basis | |
Optimizing CT protocols (Optimizing CT protocols portion was used with permission from Adam M. Alessio, PhD and Paul E. Kinahan, PhD, "CT Protocol Selection in PET-CT Imaging" Image Wisely, 2012.")
The computed tomography (CT) acquisition during PET/CT imaging is often performed for a variety of purposes. In whole-body PET/CT imaging, the CT portion can be performed as a higher dose diagnostic quality CT with intravenous and oral contrast or as a lower dose non-contrast CT primarily for anatomic localization and attenuation correction of PET images. The CT acquisition techniques should be tailored for their intended purpose. CT imaging performed as a higher dose diagnostic quality CT is typically performed with intravenous and oral contrast and imparts more radiation dose than a lower dose CT for anatomic localization and attenuation correction. If a lower dose CT scan is performed, the acquisition technique can be reduced substantially from a higher dose CT, often by 50-80% [3, 16, 17]. For the purposes of PET attenuation correction, the CT image is used to generate a low-resolution attenuation map. Thus, CT images used for attenuation correction can be many times noisier than higher dose CT images because they will be smoothed to match the PET resolution prior to generation of PET attenuation correction factors. Table 2 summarizes the typical ranges of techniques and dosimetry for these two categories of CT imaging for adult whole-body FDG-PET/CT for a scan range from the eye to mid-thigh. Additional helpful dosimetry can also be obtained from U.S. Diagnostic Reference Levels and Achievable Doses for 10 Adult CT Examinations [18].
| TABLE 2. TYPICAL ACQUISITION TECHNIQUES USED FOR WHOLE-BODY FDG-PET-CT IMAGING | ||
| Study | Injected Activity | Effective Dose Estimate |
| PET [19, 20] | [5-15] mCi FDG Injected (185-555 MBq) |
3.5-10.5 mSv |
| Higher dose diagnostic quality CT performed with IV and oral (21) | [110-200] mAs (22) CTDIvol = [8-14] mGy |
11-20 mSv |
| Lower dose CT for anatomic localization and attenuation correction (21) | [30-60] mAs (17) CTDIvol = [2-4] mGy |
3-6 mSv |
For ease of comparison, all CT studies presented are performed with 120 kVp, pitch 1.375, 40 mm collimation, 900 mm scan range, average tube current-time product.
When higher dose diagnostic quality CT studies are prescribed in addition to a whole-body FDG-PET/CT examination, the diagnostic CT region of interest may differ from the conventional PET “Skull base-to-mid-thigh” tumor imaging. Often, the diagnostic CT study requires a limited field-of-view (for example chest only), which is a subset of the PET study. In these situations, the higher dose CT study could be performed over only the diagnostic region of interest, and then a low-dose attenuation correction study could be performed over the remaining regions to provide attenuation correction throughout the PET study. Some PET/CT scanners offer this flexibility for axially varied low-high-low CT acquisitions to lower exposure in appropriate regions. If a system does not support different acquisition techniques axially, it may still be advantageous to acquire the diagnostic CT only over the appropriate region of interest and acquire a separate CT for attenuation correction over the entire PET scan region. This is particularly true if there is patient motion, or the potential for patient motion, between the diagnostic CT scan and the PET scan. Misalignment between the CT scan used for attenuation correction and the PET scan can lead to substantial attenuation and/or scatter correction artifacts [23-25].
Contrast-enhanced CT can lead to errors in attenuation correction for the PET images. This is a result of the quantitative limitations of CT and the multi-linear scaling method for deriving attenuation maps from CT images [23]. For the purpose of detection of lesions, these errors are arguably minor [26, 27]. When quantification is of concern, particularly for therapy assessment and clinical trials, correction steps are needed to account for presence of contrast material [28]. Many commercial systems offer corrections to reduce these errors, although they can lead to increased errors in other highly attenuating regions, such as bone, and at lower kVp settings [29].
Reduction of dose from the CT component of PET/CT may be undertaken in various ways. The attenuation-correction CT scan typically extends from the skull base to the proximal thighs. However, the anatomical extent may be reduced further in certain situations. For example, elimination of imaging of the pelvis for lesions such as head and neck cancer or other primary tumors that do not frequently spread to the pelvis may reduce dose, although more data is needed before adopting this type of strategy. Newer reconstruction techniques such as adaptive statistical iterative reconstruction (ASIR) and model-based iterative reconstruction (MBIR), which are being adapted increasingly, may prove more valuable in reducing dose in the future. Through use of ASIR, the dose of a higher dose diagnostic quality CT can potentially be reduced by up to 65% in adults without compromising image quality [30, 31]. Similarly, MBIR can allow for up to 80% reduction of radiation dose, although the prolonged processing time may limit its routine use in clinical practice [32, 33]
Image noise, which degrades CT image quality, is inversely related to the X-ray beam energy and increases as tube current or tube voltage decreases. The challenge to the practicing radiologist and nuclear medicine physician is to determine the acceptable range of image quality and to establish the minimum radiation doses needed to achieve this range. Many PET/CT devices default to 140 kVp for attenuation correction scans; however, reduction to 80-120 kVp can significantly reduce the dose and should be considered in adult patients [34-36]. Lower tube currents and exposure times (as low as 16-50 mAs) may also be used to decrease radiation dose while maintaining adequate image quality [37]. Automatic tube current modulation (or automatic exposure control [AEC]), which automatically adapts tube current in both angular and longitudinal directions according to patient size, can be useful to reduce patient dose exposure by 20-60% while maintaining predefined image noise or image quality characteristics [38-40]. Organ-based tube current modulation (TCM), in which tube current is decreased as the X-ray tube passes over the anterior surface of the body and increased over the posterior surface of the body, can also be implemented to decrease dose to anterior superficial radiosensitive organs such as the breast, thyroid gland, and eye lens by up to 50% without compromising image quality [41-43]. Increasing pitch has also been reported as a means to decrease CT dose exposure [44, 45]
PET/CT in Pregnant Patients
PET/CT may rarely be performed as a problem-solving diagnostic modality in specific pregnant patients where the possible clinical information obtained from the study is felt to outweigh the risks of radiation to the fetus.
Based on potential risks of fetal loss, teratogenicity, fetal growth retardation, and carcinogenesis, FDG-PET/CT is generally contraindicated in the pregnant patient. PET/CT is more likely to be performed inadvertently in patients with unsuspected pregnancy. This would most often occur in very early pregnancy, perhaps after only a few weeks gestation, especially at a time that routine urine pregnancy exams are less reliable. Occasionally, human error or miscommunication may be responsible for such events. Medical physicists, radiologists, and nuclear medicine physicians may be consulted to evaluate a case and consult with a pregnant patient before or after a planned or unplanned PET/CT examination [46-49].
For head, neck, or chest CT examinations where the fetus is not directly irradiated, maternal Compton scatter radiation doses to the fetus are on the order of 0.2 mGy or less, depending on fetal size and proximity to the primary X-ray beam. With typical abdominal-pelvic CT, the fetus is directly exposed to the scanning beam, with estimated doses of 10-25 mGy expected [50, 51]. When a risk-benefit analysis in a given clinical situation favors imaging using CT, the goal is to produce diagnostic-quality images with the minimum fetal radiation exposure.
For abdominal-pelvic CT in a pregnant patient, depending on patient size, 120 kVp or less and a pitch of greater than 1 may be prescribed [50, 51]. Tube current modulation may be used with care to avoid low noise level settings that could result in a higher fetal dose. Where available, iterative reconstruction methods should be used to improve image reconstruction quality. Multiphase imaging should be avoided.
Normalized fetal CT Monte Carlo dose estimates range from 7.3 to 14.3 mGy/100 mAs [52]. Lazarus et al. reported a mean dose of 17.1 mGy (range 8–44 mGy) [53]. In a recent series of 54 patients, investigators estimated mean fetal dose to be 24.8 mGy (range, 6.7–56 mGy) [50].
Doses to the fetus from a single-acquisition abdominal-pelvic CT examination have ranged between 10 and 50 mGy in phantom and clinical studies. The most important stochastic risk in this dose range is a potential 1.5- to 2.0-fold increased risk of childhood leukemia. This translates into a relative leukemia risk of 3 in 1000 at background radiation levels to 4 in 1000 at 10 mGy and 6 in 1000 at 50 mGy [51]. The risks of fetal adverse outcomes, including childhood cancer induction, are small at a dose of 100 mGy and negligible at doses of less than 50 mGy [51, 52, 54].
Iodinated contrast agents are known to cross the human placenta and enter the fetus [55]. While iodinated contrast agents have not been associated with teratogenic effects, and there are no reports of clinical sequelae induced by iodinated contrast agents administered intravenously, iodinated contrast agents can cause neonatal hypothyroidism if directly instilled into the amniotic fluid [56]. A recent study of the effect of in utero exposure to a single high dose of iodinated contrast material on neonatal thyroid function identified no significant risks [57]. The ACR Manual on Contrast Media recommends that iodinated contrast agents should be used only as needed in pregnant patients [55].
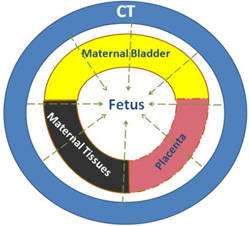
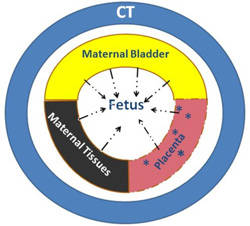
Radiotracers deliver fetal radiation by two mechanisms (Figure 1 and Table 4):
- Irradiation from radioactivity in maternal organs including bladder, placenta, and bowel.
- Internal irradiation (beta+ and gamma) from trans-placental radiotracers.
Typically, smaller moieties of 500 Daltons or less readily traverse the placental barrier. As PET radiotracers typically represent small molecules, the available PET radiotracers FDG, Na18F, 82Rb, and 18F-Florbetapir would be expected to cross the placenta and directly irradiate the fetus [58-61].
| TABLE 3 - FETAL DOSE ESTIMATED FROM MATERNAL FDG, INCLUDING PLACENTAL CROSSOVER16 | ||||
| Early | 3-Month | 6-Month | 9-Month | |
| Dose from FDG (mGy/ mCi) | 0.81 | 0.81 | 0.63 | 0.63 |
PET/CT in the Pregnant Patient: Fetal dosimetry
Total fetal estimate radiation dose from PET/CT is the sum of CT exposure + maternal gamma irradiation + fetal beta+ + fetal gamma irradiation (Figure 1). Dosimetry estimates from clinical cases are presented in Table 3. Depending on the PET and attenuation correction technique applied, fetal age, and maternal body habitus, fetal estimated doses of between 1.1 and 21.9 mGy have been reported.
| FIGURE 1 - FETAL RADIATION ESTIMATED EXPOSURE FROM 18F-FDG PET-CT | ||
 |
 |
 |
|
CT Exposure |
Maternal Tissue Irradiation |
Trans-Placental Radiation |
| TABLE 4 - FETAL ESTIMATED IRRADIATION FROM FDG-PET/CT CASE REPORTS | ||||
| Maternal | Placental Crossover | CT | Total | |
| Zanotti-Fregonara et al 2010 (60) | 3.0 mGy | 8.9 mGy | 10 mGy | 21.9 mGy |
| Takalkar et al 2011 (61) | 1.1-9.4 mGy (reported combined maternal plus placental crossover) | Negligible (68Ge rod)19 |
1.1-9.4 mGy | |
| Hsieh et al 2012 (62) | 6.3 mGy (reported combined maternal plus placental crossover) | 3.6 mGy | 9.9 mGy | |
PET/CT in the Pregnant Patient: Recommendations
- Establish presence and gestational age of pregnancy
- Establish the indication for medical imaging
- Consider non-ionizing radiation alternative imaging modalities, specifically ultrasound and MRI
- Consider feasibility of obstetrical interventions, including termination or early delivery
- If FDG-PET is indicated during pregnancy, consider where appropriate:
- FDG dose reduction to 5 mCi
- Apply PET imaging techniques such as 3D when possible to support reduced FDG dosing
- Increased oral hydration or intravenous saline
- Furosemide 20 mg IV, 15 min after FDG administration
- Encourage frequent voiding
- Selective urinary bladder catheterization
- Attenuation correction with a 68Ge rod source is most radiation efficient
- CT attenuation with 120 kVp and a pitch greater than 1
- Apply tube current modulation with care to exclude low noise settings
- Apply iterative CT reconstruction wherever possible
- Estimate total fetal dose: proposed interpretation
- Recommendations: < 1 mGy (total gestation)
- NRC worker limit: < 5 mGy (0.50 mGy/month)
- Fetal doses of < 50 mGy, no evidence for fetal injury
- Fetal doses of < 100 mGy, termination not justified
- At fetal doses between 100 and 150* mGy, consider individual circumstances
- Fetal doses > 150* mGy, possible fetal damage, termination should be seriously considered [*equivalent exposure >3 pelvic CT exams]
- Fetal doses > 500 mGy, fetal risk sufficient to recommend termination
Additional information is available in the ACR Practice Guideline for Imaging Pregnant Adolescents and Women with Ionizing Radiation.
Appropriate PET/CT imaging protocol
Careful selection of patients to be imaged and the appropriate PET/CT imaging protocol should be a priority of the radiologist/nuclear medicine physician and the referring physician in order to avoid unnecessary repeated exposure. Risk-benefit ratios of whole-body PET/CT must be carefully evaluated before each study is ordered. This is especially important in cases where the clinical utility is less well established, in addition to the younger patient population who may survive for many years after the treatment and cure of their malignancy [4, 63, 64]. Communication between the referring physician, various clinical departments, and the radiologist/nuclear medicine physician is essential to avoid unnecessary and repetitive imaging or excessive re-staging in patients who may have already been imaged previously at the same or other locations. Collaborative effort between radiologists/nuclear medicine physicians, imaging technologists, and medical physicists is also critical to ensure optimization of scanning protocols to reduce dose while maintaining image quality. Participation in dose registries (such as the American College of Radiology (ACR) Dose Index Registry (DIR)) can allow facilities to compare their CT dose indices to regional and national values for optimization of patient radiation doses for medical imaging. Use of alternative non-ionizing radiation imaging technologies (US, MRI) is also recommended whenever possible to reduce dose exposure over time [65]. Furthermore, use of PET/MRI as an alternative to PET/CT may also be feasible for certain clinical applications to reduce dose, although more research on this topic is needed.
The search for strategies in effective reduction of whole-body dose without compromising critical diagnostic information should continue to be an essential part of optimizing FDG-PET/CT imaging protocols.
References
- Delbeke D, Coleman RE, Guiberteau MJ, et al. Procedure guideline for tumor imaging with 18F-FDG PET/CT 1.0. J Nucl Med. 2006;47(5):885-95.
- Willowson KP, Bailey EA, Bailey DL. A retrospective evaluation of radiation dose associated with low dose FDG protocols in whole-body PET/CT. Australas Phys Eng Sci Med. 2012;35(1):49-53.
- Brix G, Nosske D, Lechel U. Radiation exposure of patients undergoing whole-body FDG-PET/CT examinations: an update pursuant to the new ICRP recommendations. Nuklearmedizin. 2014;53(5):217-20.
- Huang B, Law MW, Khong PL. Whole-body PET/CT scanning: estimation of radiation dose and cancer risk. Radiology. 2009;251(1):166-74.
- Wu TH, Huang YH, Lee JJ, et al. Radiation exposure during transmission measurements: comparison between CT- and germanium-based techniques with a current PET scanner. Eur J Nucl Med Mol Imaging. 2004;31(1):38-43.
- Khamwan K, Krisanachinda A, Pasawang P. The determination of patient dose from (18)F-FDG PET/CT examination. Radiat Prot Dosimetry. 2010;141(1):50-5.
- Etard C, Celier D, Roch P, Aubert B. National survey of patient doses from whole-body FDG PET-CT examinations in France in 2011. Radiat Prot Dosimetry. 2012;152(4):334-8.
- Murano T, Minamimoto R, Senda M, et al. Radiation exposure and risk-benefit analysis in cancer screening using FDG-PET: results of a Japanese nationwide survey. Ann Nucl Med. 2011;25(9):657-66.
- Masuda Y, Kondo C, Matsuo Y, Uetani M, Kusakabe K. Comparison of imaging protocols for 18F-FDG PET/CT in overweight patients: optimizing scan duration versus administered dose. J Nucl Med. 2009;50(6):844-8.
- Alessio A, Vesselle H, Lewis D, et al. Feasibility of low-dose FDG for whole-body TOF PET/CT oncologic workup. J Nucl Med. 2012;53(Supplement 1):476.
- Murray I, Kalemis A, Glennon J, et al. Time-of-flight PET/CT using low-activity protocols: potential implications for cancer therapy monitoring. Eur J Nucl Med Mol Imaging. 2010;37(9):1643-53.
- Conti M. Focus on time-of-flight PET: the benefits of improved time resolution. Eur J Nucl Med Mol Imaging. 2011;38(6):1147-57.
- Watson CC, Casey ME, Bendriem B, et al. Optimizing injected dose in clinical PET by accurately modeling the counting-rate response functions specific to individual patient scans. J Nucl Med. 2005;46(11):1825-34.
- Karp JS, Surti S, Daube-Witherspoon ME, Muehllehner G. Benefit of time-of-flight in PET: experimental and clinical results. J Nucl Med. 2008;49(3):462-70.
- Boellaard R, Delgado-Bolton R, Oyen WJ, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42(2):328-54.
- Gelfand MJ, Lemen LC. PET/CT and SPECT/CT dosimetry in children: the challenge to the pediatric imager. Semin Nucl Med. 2007;37(5):391-8.
- Alessio AM, Kinahan PE, Manchanda V, Ghioni V, Aldape L, Parisi MT. Weight-based, low-dose pediatric whole-body PET/CT protocols. J Nucl Med. 2009;50(10):1570-7.
- Kanal KM, Butler PF, Sengupta D, et al. U.S. Diagnostic Reference Levels and Achievable Doses for 10 Adult CT Examinations. Radiology. 2017:161911.
- ICRP. Radiation dose to patients from radiopharmaceuticals - Addendum 3 to ICRP Publication 53. ICRP Publication 106. Approved by the Commission in October 2007. Ann ICRP. 2008;38(1-2):1-197.
- The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Ann ICRP. 2007;37(2-4):1-332.
- Group I. CT dosimetry tool. St George's Healthcare NHS Trust, 2007. 2009.
- Brix G, Lechel U, Glatting G, et al. Radiation exposure of patients undergoing whole-body dual-modality 18F-FDG PET/CT examinations. J Nucl Med. 2005;46(4):608-13.
- Kinahan PE, Hasegawa BH, Beyer T. X-ray-based attenuation correction for positron emission tomography/computed tomography scanners. Semin Nucl Med. 2003;33(3):166-79.
- Sureshbabu W, Mawlawi O. PET/CT imaging artifacts. J Nucl Med Technol. 2005;33(3):156-61; quiz 63-4.
- Lodge MA, Mhlanga JC, Cho SY, Wahl RL. Effect of patient arm motion in whole-body PET/CT. J Nucl Med. 2011;52(12):1891-7.
- Berthelsen AK, Holm S, Loft A, et al. PET/CT with intravenous contrast can be used for PET attenuation correction in cancer patients. Eur J Nucl Med Mol Imaging. 2005;32(10):1167-75.
- Mawlawi O, Erasmus JJ, Munden RF, et al. Quantifying the effect of IV contrast media on integrated PET/CT: clinical evaluation. AJR 2006;186(2):308-19.
- Visvikis D, Costa DC, Croasdale I, et al. CT-based attenuation correction in the calculation of semi-quantitative indices of [18F]FDG uptake in PET. Eur J Nucl Med Mol Imaging. 2003;30(3):344-53.
- Abella M, Alessio AM, Mankoff DA, et al. Accuracy of CT-based attenuation correction in PET/CT bone imaging. Phys Med Biol. 2012;57(9):2477-90.
- Desai GS, Uppot RN, Yu EW, et al. Impact of iterative reconstruction on image quality and radiation dose in multidetector CT of large body size adults. Eur Radiol. 2012;22(8):1631-40.
- Hara AK, Paden RG, Silva AC, et al. Iterative reconstruction technique for reducing body radiation dose at CT: feasibility study. AJR 2009;193(3):764-71.
- Katsura M, Matsuda I, Akahane M, et al. Model-based iterative reconstruction technique for radiation dose reduction in chest CT: comparison with the adaptive statistical iterative reconstruction technique. Eur Radiol. 2012;22(8):1613-23.
- Singh S, Kalra MK, Do S, et al. Comparison of hybrid and pure iterative reconstruction techniques with conventional filtered back projection: dose reduction potential in the abdomen. J Comput Assist Tomogr. 2012;36(3):347-53.
- Gnannt R, Winklehner A, Eberli D, et al. Automated tube potential selection for standard chest and abdominal CT in follow-up patients with testicular cancer: comparison with fixed tube potential. Eur Radiol. 2012;22(9):1937-45.
- Hoang JK, Yoshizumi TT, Nguyen G, et al. Variation in tube voltage for adult neck MDCT: effect on radiation dose and image quality. AJR 2012;198(3):621-7.
- Kaza RK, Platt JF, Al-Hawary MM, et al. CT enterography at 80 kVp with adaptive statistical iterative reconstruction versus at 120 kVp with standard reconstruction: image quality, diagnostic adequacy, and dose reduction. AJR 2012;198(5):1084-92.
- Kumar S, Pandey AK, Sharma P, et al. Optimization of the CT acquisition protocol to reduce patient dose without compromising the diagnostic quality for PET-CT: a phantom study. Nucl Med Commun. 2012;33(2):164-70.
- Soderberg M, Gunnarsson M. Automatic exposure control in computed tomography--an evaluation of systems from different manufacturers. Acta Radiol. 2010;51(6):625-34.
- Jackson J, Pan T, Tonkopi E, Swanston N, Macapinlac HA, Rohren EM. Implementation of automated tube current modulation in PET/CT: prospective selection of a noise index and retrospective patient analysis to ensure image quality. J Nucl Med Technol. 2011;39(2):83-90.
- Tack D, De Maertelaer V, Gevenois PA. Dose reduction in multidetector CT using attenuation-based online tube current modulation. AJR 2003;181(2):331-4.
- Duan X, Wang J, Christner JA, et al. Dose reduction to anterior surfaces with organ-based tube-current modulation: evaluation of performance in a phantom study. AJR 2011;197(3):689-95.
- Wang J, Duan X, Christner JA, et al. Bismuth shielding, organ-based tube current modulation, and global reduction of tube current for dose reduction to the eye at head CT. Radiology 2012;262(1):191-8.
- Hoang JK, Yoshizumi TT, Choudhury KR, et al. Organ-based dose current modulation and thyroid shields: techniques of radiation dose reduction for neck CT. AJR 2012;198(5):1132-8.
- Ketelsen D, Buchgeister M, Korn A, et al. High-pitch computed tomography coronary angiography-a new dose-saving algorithm: estimation of radiation exposure. Radiol Res Pract 2012;2012:724129.
- Achenbach S, Marwan M, Schepis T, et al. High-pitch spiral acquisition: a new scan mode for coronary CT angiography. J Cardiovasc Comput Tomogr. 2009;3(2):117-21.
- Wieseler KM, Bhargava P, Kanal KM, et al. Imaging in pregnant patients: examination appropriateness. Radiographics 2010;30(5):1215-29; discussion 30-3.
- Williams PM, Fletcher S. Health effects of prenatal radiation exposure. Am Fam Physician 2010;82(5):488-93.
- Patel SJ, Reede DL, Katz DS, et al. Imaging the pregnant patient for nonobstetric conditions: algorithms and radiation dose considerations. Radiographics 2007;27(6):1705-22.
- McCollough CH, Schueler BA, Atwell TD, et al. Radiation exposure and pregnancy: when should we be concerned? Radiographics 2007;27(4):909-17; discussion 17-8.
- Goldberg-Stein S, Liu B, Hahn PF, Lee SI. Body CT during pregnancy: utilization trends, examination indications, and fetal radiation doses. AJR 2011;196(1):146-51.
- Goldberg-Stein SA, Liu B, Hahn PF, Lee SI. Radiation dose management: part 2, estimating fetal radiation risk from CT during pregnancy. AJR 2012;198(4):W352-6.
- Ratnapalan S, Bona N, Chandra K, Koren G. Physicians' perceptions of teratogenic risk associated with radiography and CT during early pregnancy. AJR Am J Roentgenol. 2004;182(5):1107-9.
- Lazarus E, Debenedectis C, North D, et al. Utilization of imaging in pregnant patients: 10-year review of 5270 examinations in 3285 patients--1997-2006. Radiology. 2009;251(2):517-24.
- Y B. Ionizing and nonionizing radiation in pregnancy. In: Koren G, ed. Maternal–fetal toxicology: a clinician’s guide, 3rd ed. New York, NY: Marcel Dekker, 2001:603–651.\
- American College of Radiology (ACR) Website. ACR manual on contrast media v. 2016.
- Webb JA, Thomsen HS, Morcos SK, Members of Contrast Media Safety Committee of European Society of Urogenital R. The use of iodinated and gadolinium contrast media during pregnancy and lactation. Eur Radiol 2005;15(6):1234-40.
- Bourjeily G, Chalhoub M, Phornphutkul C, et al. Neonatal thyroid function: effect of a single exposure to iodinated contrast medium in utero. Radiology 2010;256(3):744-50.
- Russell JR, Stabin MG, Sparks RB. Placental transfer of radiopharmaceuticals and dosimetry in pregnancy. Health Phys 1997;73(5):747-55.
- Stabin MG. Proposed addendum to previously published fetal dose estimate tables for 18F-FDG. J Nucl Med 2004;45(4):634-5.
- Zanotti-Fregonara P, Jan S, Taieb D, et al. Absorbed 18F-FDG dose to the fetus during early pregnancy. J Nucl Med 2010;51(5):803-5.
- Takalkar AM, Khandelwal A, Lokitz S,et al. 18F-FDG PET in pregnancy and fetal radiation dose estimates. J Nucl Med. 2011;52(7):1035-40.
- Hsieh TC, Wu YC, Sun SS, et al. FDG PET/CT of a late-term pregnant woman with breast cancer. Clin Nucl Med. 2012;37(5):489-91.
- Petrausch U, Samaras P, Haile SR, et al. Risk-adapted FDG-PET/CT-based follow-up in patients with diffuse large B-cell lymphoma after first-line therapy. Ann Oncol. 2010;21(8):1694-8.
- Hardin LV Ravenel J, Gordon L, et al. Radiation risks to lymphoma patients undergoing 18F-FDG studies. J Nucl Med. 2009;50 (Supplement2):1380.
- Semelka RC, Armao DM, Elias J, Jr., Huda W. Imaging strategies to reduce the risk of radiation in CT studies, including selective substitution with MRI. J Magn Reson Imaging. 2007;25(5):900-9.



